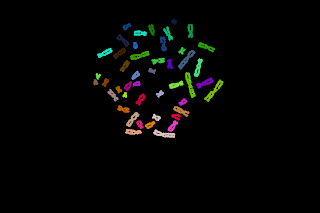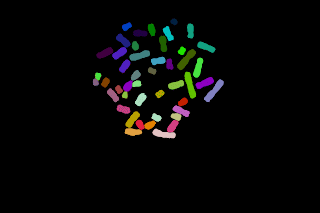In a previous post, I was wondering how to label the chromatids of metaphasic chromosomes. The idea was to use connected components obtained after high pass filtering, then to assign those region to one chromatid or the other one.
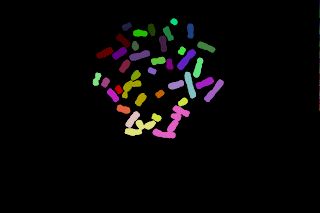
Here, I believe it should be possible to proceed with pre-labelled seeds and let them grow inside a mask as shown in the following images:
The top left image is the seeds image and the top right one is the mask image. The seeds are allowed to grow only in the white region of the mask image, so this operation is a geodesic transformation.
- Depending of the usage, different gray-scaled seeds can be designed. For example the big light-blue/magenta one could be used to label the left side and the right side of the large white particle. The smaller blue/yellow seed could be used to label the upper or the lower part of a chromosome like particle.
- Naively if gray-scale dilation followed by masking is performed on the image seeds, the lower left image is obtained, whereas a result similar to the lower right image was waited. A simple gray-scale dilation yields to imbalanced "blue/pink" regions.